A positive control validates the proper storage of reagents and the consistent performance of the instrument. Depending on the type of positive control, it can also validate the success of the sample preparation as well as the quality of the specimen. Ultimately, the positive control validates the amplification itself.
PCR is the most commonly used analysis in laboratories to detect the DNA of infectious agents. with this type of analysis, an exogenous positive control is often used, meaning that a synthetic DNA (which does not originate from the animal) is added to the reaction to be detected by the control test. This type of positive control validates the proper storage of the reagents, the consistent performance of the instrument and even the sample preparation, however, it does not ensure that specimen collection has been carried out properly. There is no guarantee that the pathogen, if present, has been properly collected. If the tested sample is negative and its control is positive, the specialized laboratory cannot determine if the result is negative because the animal is not infected or if the specimen was not properly collected. In this case, it is a potential false-negative.
Epona tests are developed with an endogenous positive control, in other words, a control test aimed at detecting the DNA of the animal itself, via cells collected by nasopharyngeal swab. In addition to providing evidence of proper reagent storage, instrument performance, and proper sample preparation, the control also demonstrates that the swab carries adequate biological material from the nasopharynx.
This positive control provides an additional quality control, such that if a appears as + the result of the positive control, it could indicate improper specimen collection. To obtain the best specimen, it’s essential to use the proper material and technique.
The type of cotton swab used and the technical procedure of specimen collection are essential for the optimal sensitivity of molecular tests (PCR or Epona tests). Omitting either one of those two aspects can lead to an elevated number of false negatives. These errors are detectable by an endogenous positive control, such as that used in the Epona tests.
The use of an endogenous positive control such as that used in the Epona tests has demonstrated that nasopharyngeal swabs with a large cotton tip (approximately 1 cm in diameter) provide the best quality in terms of specimen collection, given that their length reaches the nasopharynx (see image).
Swabs with a finer cotton tip, designated for genital specimens can result in inadequate collection of biological material. This has a negative effect on the overall sensitivity of molecular biology tests (PCR and Epona tests), and thus the thin swab should not be used for this type of sampling.
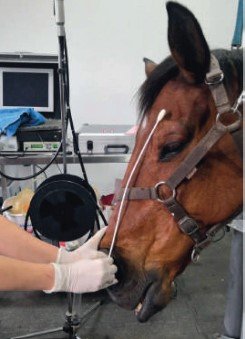
In addition to the type of swabs, the swabbing technique is important. The filthy appearance of the swab is not a reliable indicator of proper sampling. Dust and nasal secretions are often collected before essential biological material. With the nasopharynx just below the horse’s eyes, almost the entirety of the swab should be inside the nasal cavity during swabbing.

